 
With this information and Thomson’s mass-to-charge ratio, Millikan determined the mass of an electron: Subsequently, the American scientist Robert Millikan (1868–1953) carried out a series of experiments using electrically charged oil droplets, which allowed him to calculate the charge on a single electron. Another set of electrode plates deflect the ray, with the ray bending towards the positive plate. Schematic of cathode ray tube with deflection. Image used with Permission (CC BY-SA-NC). As the cathode rays travel toward the right, they are deflected toward the positive electrode (+), demonstrating that they are negatively charged. In his honor, the element Rutherfordium (number 104) was named after him.\): Deflection of Cathode Rays by an Electric Field. The nucleus has changed for the world, for good, and for bad. We can also create theories off this discovery and build on its concepts. It has allowed us to gather a deeper understanding of the shape and structure of an atom. This discovery has laid the foundations for the modern subjects of chemistry and physics. 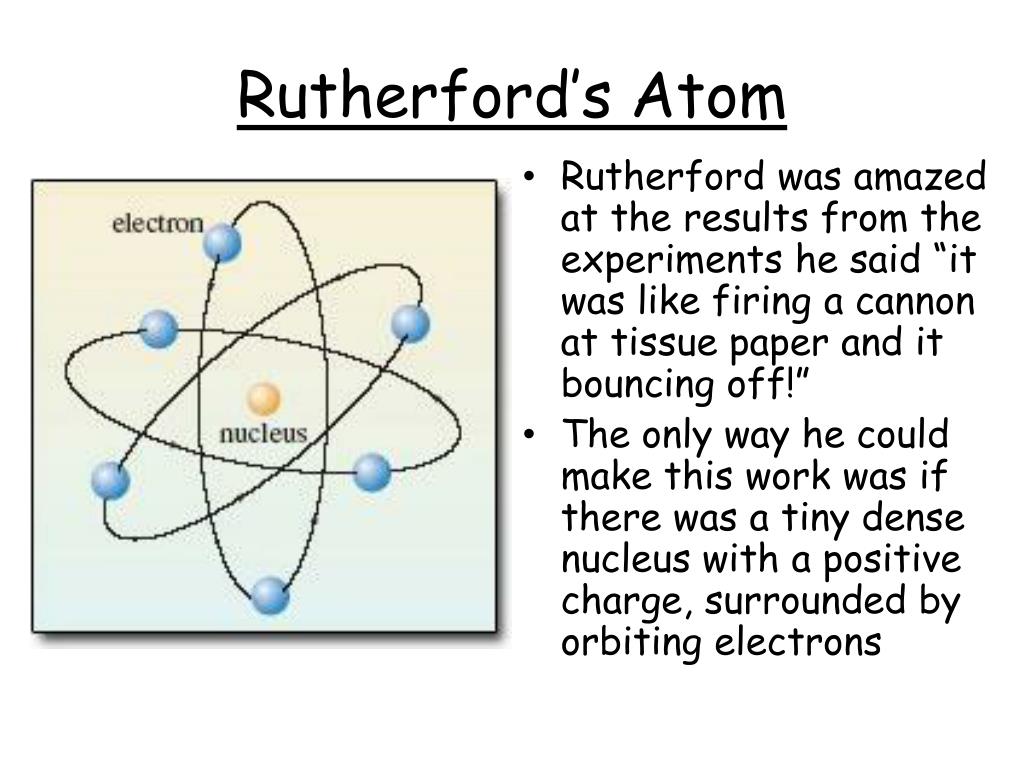
Rutherford is considered one of the greatest scientists in history because of his work with the structure of the atom. This chain reaction goes on and on, killing thousands of living organisms. These released neutrons then split more atoms. The nuclear bomb relies on a small particle (neutron) splitting the nucleus of an atom and releasing neutrons. The types of technological and other advancements that have occurred and impacted our lives both positively and negatively since Rutherford’s discoveries are seemingly endless however none may be more significant than the development of the most destructive weapon known to mankind, the nuclear bomb. His work was fundamental to quantum mechanics which explains how atoms can exist in multiple states at once. This has allowed us to classify and gain a deeper understanding of all the elements of the periodic table and we can understand the ways atoms and molecules react with each other in equations. His discoveries of the nucleus which led to the discovery of the structure of the atom underpin the entire foundation of modern chemistry today. Rutherford’s work laid the foundations of modern chemistry today. The experiment detects this scattering of alpha particles by having a fluorescent screen surrounding the setup that fluoresces when an alpha particle hits it. The alpha particles go straight through the empty space but when they come into contact with the nucleus of one of these atoms, the alpha particles bounce in a weird direction. The experiment creates a stream of alpha particles from a sample of radium in a lead cube, directed at a thin Gold or Platinum sheet 70-100 atoms in thickness. They then created the Geiger-Marsden experiment, also known as the gold foil experiment, to study the scattering of Alpha particles. In 1910, he handed his findings to his apprentices, Ernest Marsden and Hans Geiger. This is where he then studied emanations of Radium which produced a constant stream of alpha particles. He then published his findings and moved to Manchester as a result. 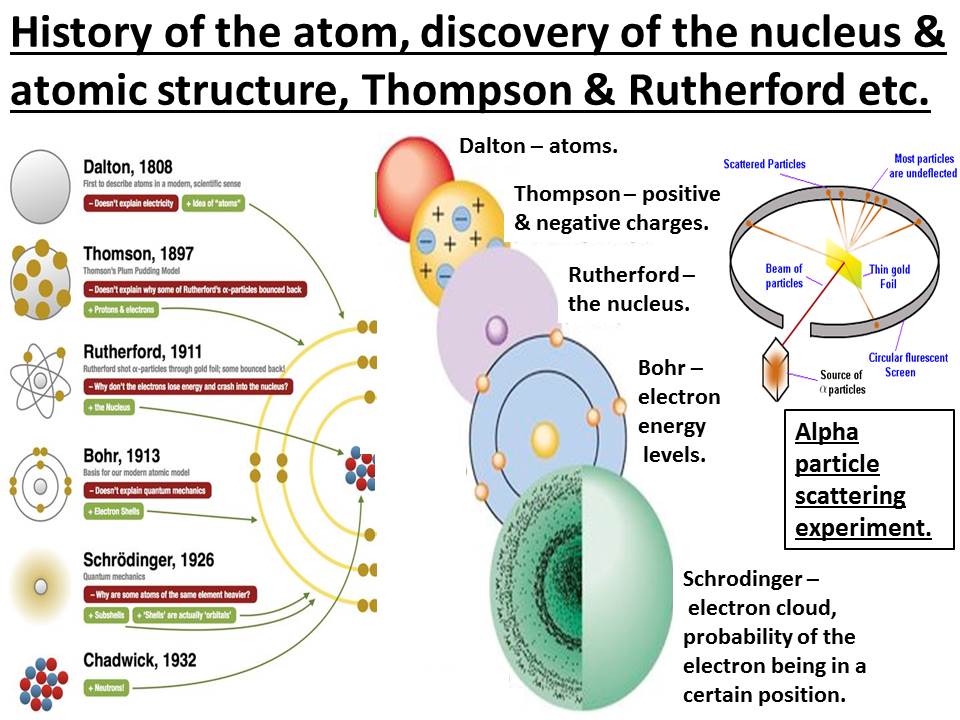
He was studying emanations of Thorium with R.D Owens when he came across Thoron, an isotope of Radon. In Montreal, Ernest Rutherford continued his work on Alpha rays. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
